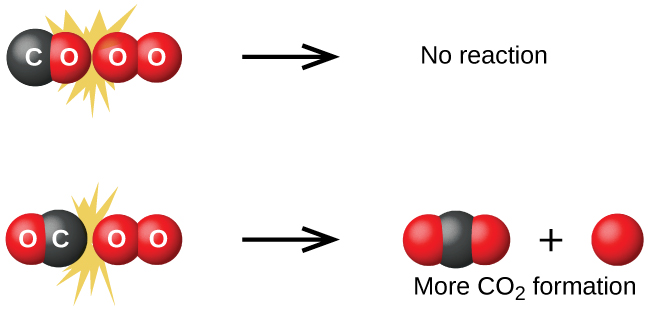
Along with that, we will briefly overview some scenarios involving nonreactive collisions. Our main goal is to define the concept of activation energy and explain why some of the collisions do not result in a reaction.Īccordingly, this article will primarily focus on the concept of reactive collision. Instead, if the colliding particles do not have sufficient kinetic energy or proper orientation in space, the reaction will not occur.īut what is meant by “sufficient energy” or “proper orientation”? To get a better idea of the concepts mentioned earlier, the article will provide some general definitions accompanied by corresponding examples and sample problems. On the other hand, not all collisions promote the reaction to take place. One of such variables is considered to be a collision. Keep reading to learn more about collision theory.Įvery chemical reaction requires collisions between the reactant particles (atoms and/or molecules). 
Furthermore, there are several variables that can also impact the rate of a reaction (concentration, temperature, catalyst, etc.). One of the most important factors that affect the speed of a reaction is the reactivity of chemicals involved in that specific reaction. The study of kinetics enables scientists to determine the relationship between the changes in conditions and the speed of a particular chemical reaction. What is meant by proper orientation in a reaction?.Why is the energy required for a reaction to take place?.What are the three parts of collision theory?. 
As you can see above, the reaction proceeds in 2 steps.To plot the activation energy graph, it is crucial to consider two variables:.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
